# Triage
**Criteria for Testing**
Testing capacity has been in flux (see [Module 3: Testing Capacity](https://curriculum.covidstudentresponse.org/module-3-current-situation-and-healthcare-response/testing-capacity-and-eligibility) for updates), with resultant changes in testing eligibility protocols. The Massachusetts Department of Public Health (MA DPH) publishes updated guidelines with criteria for COVID-19 testing (see below for version as of 4/2/2020). It differentiates between populations recommended to be tested at state versus commercial laboratories.
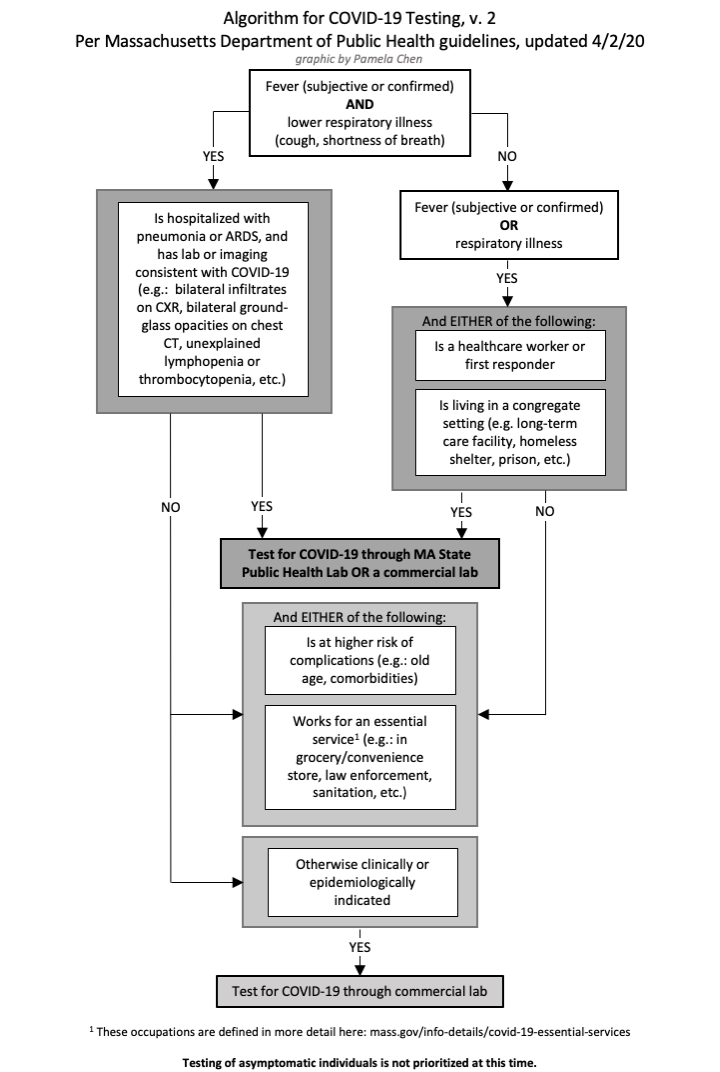
### **Remote Screening**
Screening can occur remotely through a telephone/virtual visit by guidelines similar to above. Drive-through testing sites are expanding across the country, [including Massachusetts](https://cvshealth.com/newsroom/press-releases/cvs-health-expands-rapid-covid-19-drive-through-testing-sites-massachusetts), and often require that patients fill out [an online screening assessment](https://www.cvs.com/minuteclinic/covid-19-testing) to determine eligibility. With in-house testing increasing at hospitals, institutions are developing their own testing eligibility protocols (see below). The Cleveland Clinic has produced [an online tool](https://my.clevelandclinic.org/landing/preparing-for-coronavirus?utm_campaign=coronavirus-url\&utm_medium=offline\&utm_source=redirect\&utm_content=coronavirus-url&_ga=2.212126743.2068788212.1584840182-1300742023.1582059042) allowing patients to self-assess for infection risk, with care recommendations based on risk level.
### Triage Guidelines by Hospital
Testing: Hospitals have generally been outlining testing criteria for ambulatory vs emergency department or inpatient settings. Partners-specific guidelines are available [here](https://pulse.partners.org/hub/departments/emergency_preparedness/coronavirus) (note: requires Partners credentials). BILH [provides guidelines on their website.](https://covid-19.bilh.org/covid-19-materials/) Cambridge Health Alliance guidelines are available [here](https://sites.google.com/challiance.org/ncov-quick-guide/home) (note: requires CHA credentials). Of note, most protocols are initially symptom-based (except for select populations such as transplant patients or requiring urgent airway surgery), followed by a prioritization list for symptomatic patients or staff. These reflect a balance of clinical/operational needs for testing and resource availability.
Clinical Triage: For a discussion of clinical triage guidelines, see [Module 1: Triage Guidelines. ](https://curriculum.covidstudentresponse.org/module-1-from-bench-to-bedside/management-of-covid-19#triage-guidelines)
*Thought question:*
For a low-risk patient who may not fall under the recommendations to get tested for COVID-19, but who does want to get tested, how might a healthcare worker navigate the conversation about the utility of testing amongst those at highest risk?
##
---
# Agent Instructions: Querying This Documentation
If you need additional information that is not directly available in this page, you can query the documentation dynamically by asking a question.
Perform an HTTP GET request on the current page URL with the `ask` query parameter:
```
GET https://curriculum.covidstudentresponse.org/module-6-training-for-clinical-roles/triage.md?ask=
```
The question should be specific, self-contained, and written in natural language.
The response will contain a direct answer to the question and relevant excerpts and sources from the documentation.
Use this mechanism when the answer is not explicitly present in the current page, you need clarification or additional context, or you want to retrieve related documentation sections.